It is necessary to periodically remove some of the impure electrolyte and to top it up with clean electrolyte containing high concentrations of dissolved silver. The anode compartment must be isolated from the cathode compartment by a membrane or porous bag to avoid contaminating the silver crystals with anode slime.īecause the anodes are impure and the cathodic reduction is selective for silver, dissolved copper and other base metals continually build up in solution as the silver content of the electrolyte is depleted. The production in the anode compartment of a gold-bearing slime, which can cause passivation of the silver anodes, must be periodically removed, creating materials-handling challenges. Metals such as gold and platinum, which are insoluble in nitric acid, do not dissolve and report to the anode mud. 
The main contaminant in the anode is copper, with minor amounts of lead, cadmium, and zinc. Under ideal conditions, the cathodic process selectively reduces silver, but in practice the extent of selectivity is dependent on the solution composition, applied voltage, and current density.Īnodic dissolution of the impure silver is not selective, and any readily oxidizable metallic impurity in the anode will dissolve. Additives, such as tartaric acid, can be added to control the morphology of silver crystal product. The silver crystals fall to the bottom of the electrolytic cell and are harvested, manually or automatically, on a continuous or batch basis. Silver is produced as fine needle-like crystals at the cathode, which are continually scraped off to avoid short-circuiting between the anode and cathode. 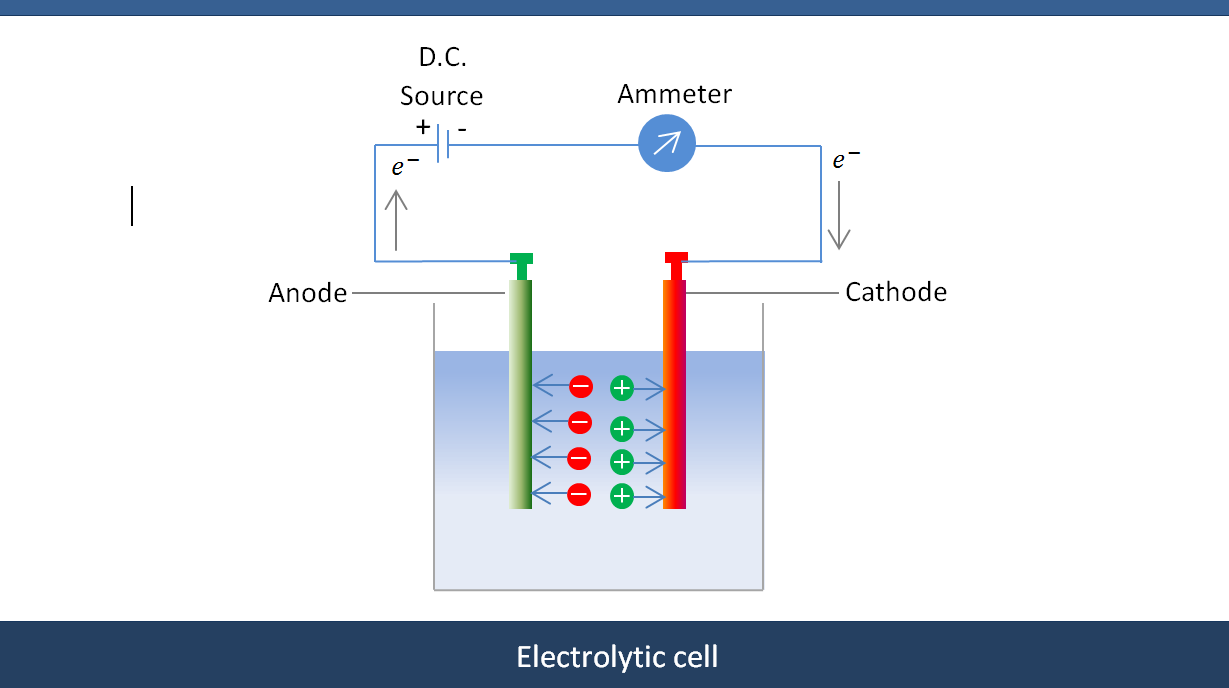
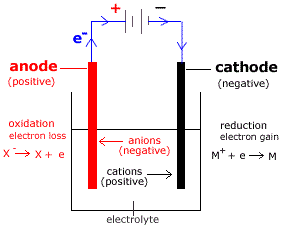
The atoms join up in pairs to form Cl 2 molecules, so chlorine gas is formed at the positive electrode.(34.21) Cathodic reduction : Ag + + e → Ag o Here, they lose electrons to form chlorine atoms. Negatively charged chloride ions move to the positive electrode. Here, they gain electrons to form calcium atoms, so calcium is formed at the negative electrode. Positively charged calcium ions move to the negative electrode. Predict the products of electrolysis of molten calcium chloride. Lead and bromine form during the electrolysis of molten lead bromide Example So lead forms at the negative electrode and bromine forms at the positive electrode.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
